Search Results
Results for: 'F.1 % change'
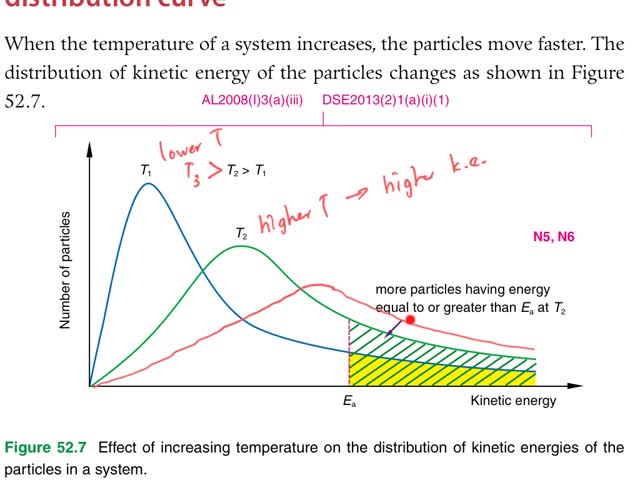
Chemistry tutorial-Ch52-4-Explaining effect of temperature change on reaction rate
By: t0605, Views: 1504
Chemistry tutorial-Ch52-4-Explaining effect of temperature change on reaction rate
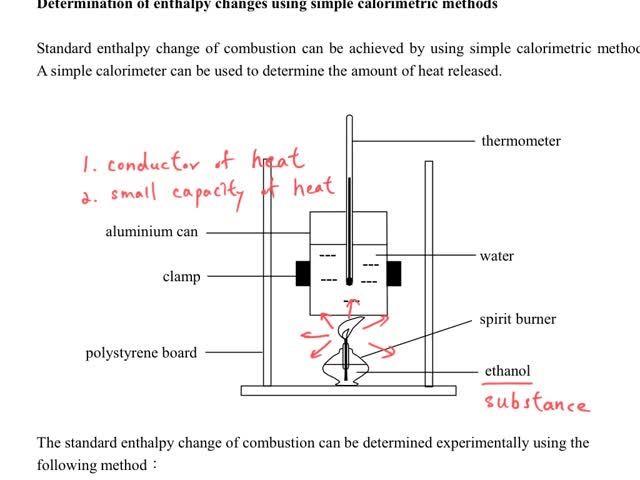
Chemistry tutorial-Ch34-6-Calorimetric method for measuring standard enthalpy change of combustion
By: t0605, Views: 1941
Chemistry tutorial-Ch34-6-Calorimetric method for measuring standard enthalpy change of combustion
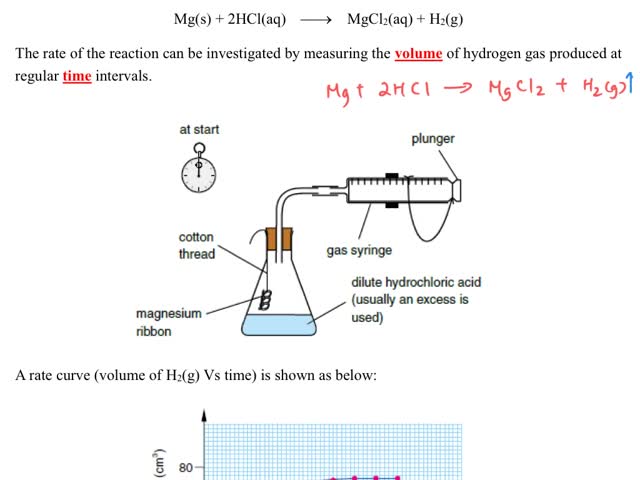
Chemistry tutotial-Ch36-3-Follow the progress of reaction by measuing the change in volume of gases
By: t0605, Views: 1735
Chemistry tutotial-Ch36-3-Follow the progress of reaction by measuing the change in volume of gases
By: t0605, Views: 1766
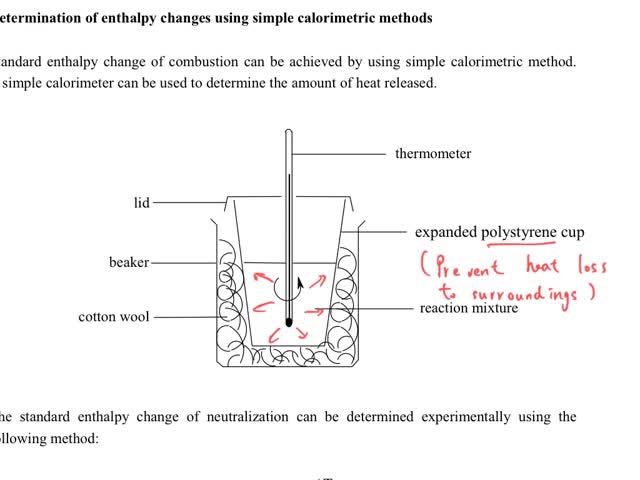
Chemistry tutorial-Ch34-8-Calorimetric method for measuring standard enthalpy change of neutralization
By: t0605, Views: 1718
Chemistry tutorial-Ch34-9-Affect of the strength of acids or bases on the standard enthalpy change of neutralization
Chemistry tutorial-Ch34-1-Standard conditions of standard enthalpy change
By: t0605, Views: 3893
Chemistry tutorial-Ch34-1-Standard conditions of standard enthalpy change
By: t0605, Views: 1809
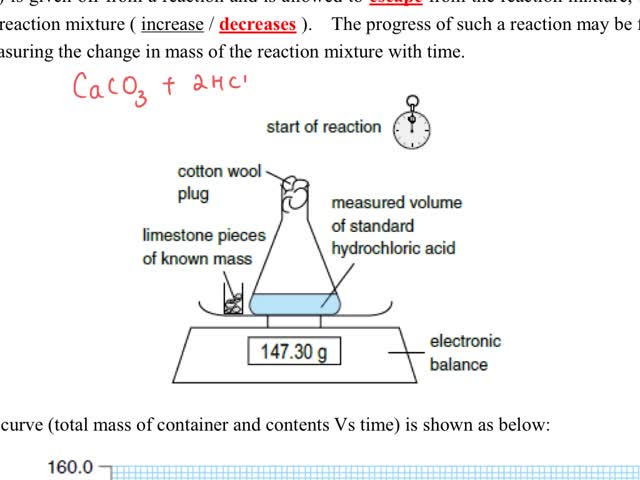
Chemistry tutotial-Ch36-5-Follow the progress of reaction by measuing the change in mass of reaction mixture
By: t0605, Views: 1656
Chemistry tutotial-Ch36-4-Follow the progress of reaction by measuing the change in pressure of reaction mixture
By: t0605, Views: 1330
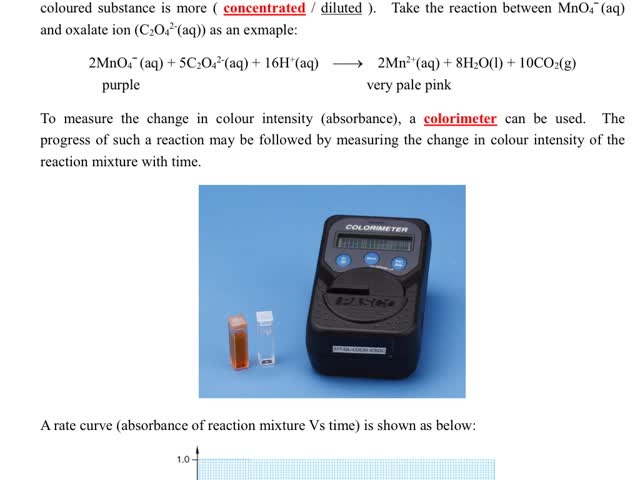
Chemistry tutotial-Ch36-6-Follow the progress of reaction by measuing the change in colour intensity of reaction mixture
YU CHUN KEUNG MEMORIAL COLLEGE